
A clinical data registry is an organized system that collects uniform data (clinical and patient-reported) to evaluate specified outcomes for a population based on performance measures developed from Clinical Practice Guidelines.
With the increasing usage of electronic health record (EHR) systems, registries have emerged as valuable solutions to harness the power of information technology. Registries can capture statistically relevant, evidence-based data to aid in decisions regarding the most optimal patient care. Other medical specialties and medical associations, including the American College of Cardiology and the American Academy of Ophthalmology, have developed their own registries.
Based on the data abstracted from the practice’s EHR, quality metrics are populated and displayed on a dashboard. Where possible, Reg-ent schedules automatic extractions of this data (more below). The data received is mapped to the measure’s “data dictionary.” For each measure, there are specific codes and keywords to identify eligible encounters, such as patient’s diagnosis, procedures, age, etc. For encounters that qualify, the defined quality action is identified for the clinicians through codes and keywords in the data dictionary.
The performance calculated from the measure logic is visualized on the dashboard. Participating clinicians may review their performance on the dashboard, compare to benchmarks, and uncover potential areas for quality improvement. Users can drill down to a specific location or physician, and track trends over time. 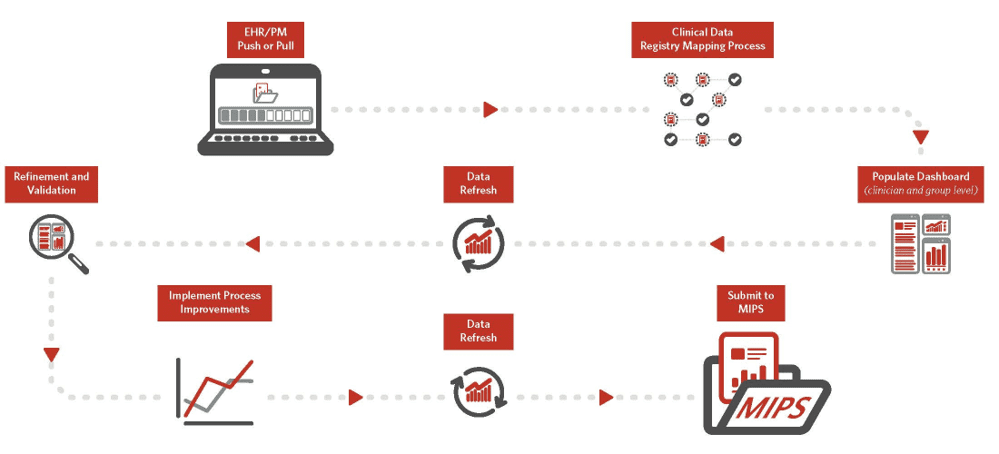
Yes, Reg-ent participants must have an EHR system to participate in Reg-ent services. This includes using Reg-ent for Merit-based Incentive Payment System (MIPS) reporting.
Reg-ent currently works with about 25 EHRs and is launching pilots with additional EHRs. Please view here for more information on integrated EHRs.
Reg-ent partners with a technical vendor, Matrix PPA, to receive data and populate the quality dashboard. Reg-ent receives practice data in two ways depending on the hosting situation of EHR.
- Data Pull is an automated approach available for practices that locally host their EHRs.
- Data Push requires practices or EHR vendors to securely share relevant data files with Matrix PPA. This approach applies to practices with cloud hosted EHRs, or practices with locally hosted EHRs that do not want to install a connector. Pushing data requires active engagement from the practice and/or EHR vendor. Some EHRs charge additional fees for this data sharing
Reg-ent is focused on specific measures and patient data only for those patients meeting the registry measures criteria. Therefore, the report run time is much quicker than that of an EHR, whose reporting functions are poorly developed and often cumbersome to use. Reg-ent offers 37 quality measures, including 11 otolaryngology-specific measures developed by the Academy and available exclusively only in Reg-ent.
Reg-ent is also the MIPS reporting tool of choice for most of our participating practices. Over the coming years, CMS plans to phase out traditional MIPS reporting for MIPS Value Pathways (MVPs). With MVPs, practices will report on a focused, subspecialty-specific measure set.
The Reg-ent team at the AAO-HNSF has codeveloped with CMS the MIPS Value Pathway focused on Otolaryngology, “Quality Care for Ear, Nose, and Throat” which was finalized as an approved MVP as outlined in the 2024 Medicare Physician Fee Schedule Final Rule. Reg-ent also provides detailed knowledge of the program and measures to assist in meaningful measure selection. You can learn more about the OTO MVP here.
Reg-Ent’s enhanced technology platform accommodates reporting for all required MIPS categories: Quality, Promoting Interoperability, and Improvement Activities. Reg-ent participants are able to view and track their scores in the Reg-ent MIPS dashboard. CMS determines final payment adjustments and shares updates directly with MIPS participants.
Reg-ent staff can also provide overviews of MIPS reporting requirements. To learn more about how Reg-ent can help your practice with the MIPS reporting, please click here.
CMS determines final payment adjustments and shares updates directly with MIPS participants.
Reg-ent participants are able to view and track their scores in each reporting category within the Reg-ent MIPS dashboard.
All final payment adjustments are determined by the Centers for Medicare and Medicaid Services (CMS) and will be applied in 2026 for the 2024 reporting year.
Please visit the Reg-ent Fees page to learn more.
Contact [email protected] with pricing or enrollment questions.
Visit the Reg-ent sign-up portal to: https://regent.entnet.org/Signup/registry.aspx.
Make sure you have the following information at hand:
- For participating clinicians:
- AAO-HNS member ID numbers of otolaryngologists and allergists
- National Provider Identifier (NPI) numbers
- Email addresses
- The practice Tax Identification Number (TIN)
- Practice location address(es)
- Credit card or check details
Upon completing the sign-up process, we begin the data sharing process. For “pull” practices, our technical vendor begins the data extraction from practice’s EHR. For “push” practices, the EHR vendor shares data files securely with our technical vendor. The data received is mapped to the measure’s “data dictionary.” For each measure, there are specific codes and keywords to identify eligible encounters, such as patient’s diagnosis, procedures, age, etc. For encounters that qualify, the quality action is identified for the clinicians through codes and keywords in the data dictionary.
The performance calculated from the measure logic is visualized on the dashboard.
The initial performance generated in your practice dashboard may show poor scores on some measures. This is often due to inconsistent data capture (not captured well, systematically, or put in the right place or right way), poor attribution (not clear which patients are in who’s panel), lack of data capture (how lab results are reported), or lack of patient panel management processes (no mechanism to identify patients who have not had immunizations, lab work, or a visit within prescribed time).
This is not unusual and is the reason why multiple mapping refinement calls are conducted. The goal is to assure better data capture, patient attribution and patient management.
You will be able to monitor patient-level quality measures, track interventions, and evaluate outcomes at a population level.
Reg-ent collects, stores and reports data on an otolaryngologist-head and neck surgeon’s behalf, taking every measure possible to safeguard it.
The AAO-HNSF is working to develop more performance measures to track both the otolaryngology patient population as well as to support the depth and breadth of the specialty. Data will be used to formulate and test the measures developed.
We will also be creating quality benchmarks so that you can compare your outcomes, and study whether or not quality improvement is necessary to improve quality across the specialty. We will also engage in research, identify gaps in care to fuel the development of additional measures and to support testing Clinical Practice Guidelines. Reg-ent has launched research opportunities for our participants with our de-deidentified data sets.
The aim of Reg-ent is to ease burden. It is designed to require minimal effort from your staff. EHR integration typically requires one to two hours per week for three to four weeks from IT staff to assist in the installation and mapping process.
Following installation, no additional work is required unless the practice’s EHR software is updated or changed, which could require repeat of the installation and mapping process. We encourage practices to review their dashboard performance with the Academy staff at least once every quarter, but the clinicians can review and track their scores based on their convenience as well.
To participate in Reg-ent, all otolaryngologists and allergists must be current members of the AAO-HNS. Each Reg-ent applicant will be validated for membership during the sign-on process. If you have any questions regarding membership status, please email the AAO-HNS Membership Services department at [email protected].
Non-physician providers such as nurse practitioners, physician assistants and speech-language pathologists are not required to become AAO-HNS members to participate in Reg-ent.
All U.S.-based practicing physicians are welcome to join Reg-ent. Physicians who are not otolaryngologists, head and neck surgeons are welcome to join AAO-HNS as Associate Members. If you have any questions regarding membership status, please email the AAO-HNS Membership Services department at [email protected].
Yes, Any U.S.-based practicing Member of the AAO-HNS, regardless of practice setting, can participate in Reg-ent. More information on Reg-ent for Academic Medical Centers can be found here.
Reg-ent has launched research opportunities for our members using the de-deidentified data sets. As more of the data completes the curation process, Reg-ent members will be able to participate in revenue generating projects as well as clinical trial research opportunities. AAO-HNS committees will also be able to request Reg-ent data for their projects. Reg-ent’s analytics partner, OM1, will offer a ‘menu’ of analytic support for those organizations that have limited data processing capabilities (i.e., Python, SQL, etc.).
Reg-ent research requests will be reviewed and prioritized by the Reg-ent Research Advisory Group (RRAG). The RRAG’s charge is to promote the highest quality internal, non-commercial Reg-ent-associated research to advance the field of otolaryngology-head and neck surgery and help define quality care within the specialty.
More information on Reg-ent Research regarding requesting data sets, eligibility, policies and procedures can be found on our website here.
Future modules beyond MIPS reporting are already under consideration including research, Maintenance of Certification, and device surveillance.
